#BlackInPhysics Celebrating Black physicists and revealing a more complete picture of what a physicist looks like.Artificial intelligence Explore the ways in which today’s world relies on AI, and ponder how this technology might shape the world of tomorrow.Collections Explore special collections that bring together our best content on trending topics.Innovation showcases A round-up of the latest innovation from our corporate partners.Projects and facilities Follow the latest progress at the world’s top scientific experiments.Policy and funding Understand how emerging policy changes could affect your work and career.Education and outreach Learn about novel approaches to educating and inspiring the scientists of the future.Business and innovation Find out how recent scientific breakthroughs are driving business innovation and commercial growth.Impact Explore the value of scientific research for industry, the economy and society.Personalities Find out who’s doing what in industry and academia.Interviews Discover the views of leading figures in the scientific community.Opinion and reviews Find out whether you agree with our expert commentators.Careers Consider your career options with valuable advice and insightful case studies.People Meet the people behind the science.Events Plan the meetings and conferences you want to attend with our comprehensive events calendar.Blog Enjoy a more personal take on the key events in and around science. 
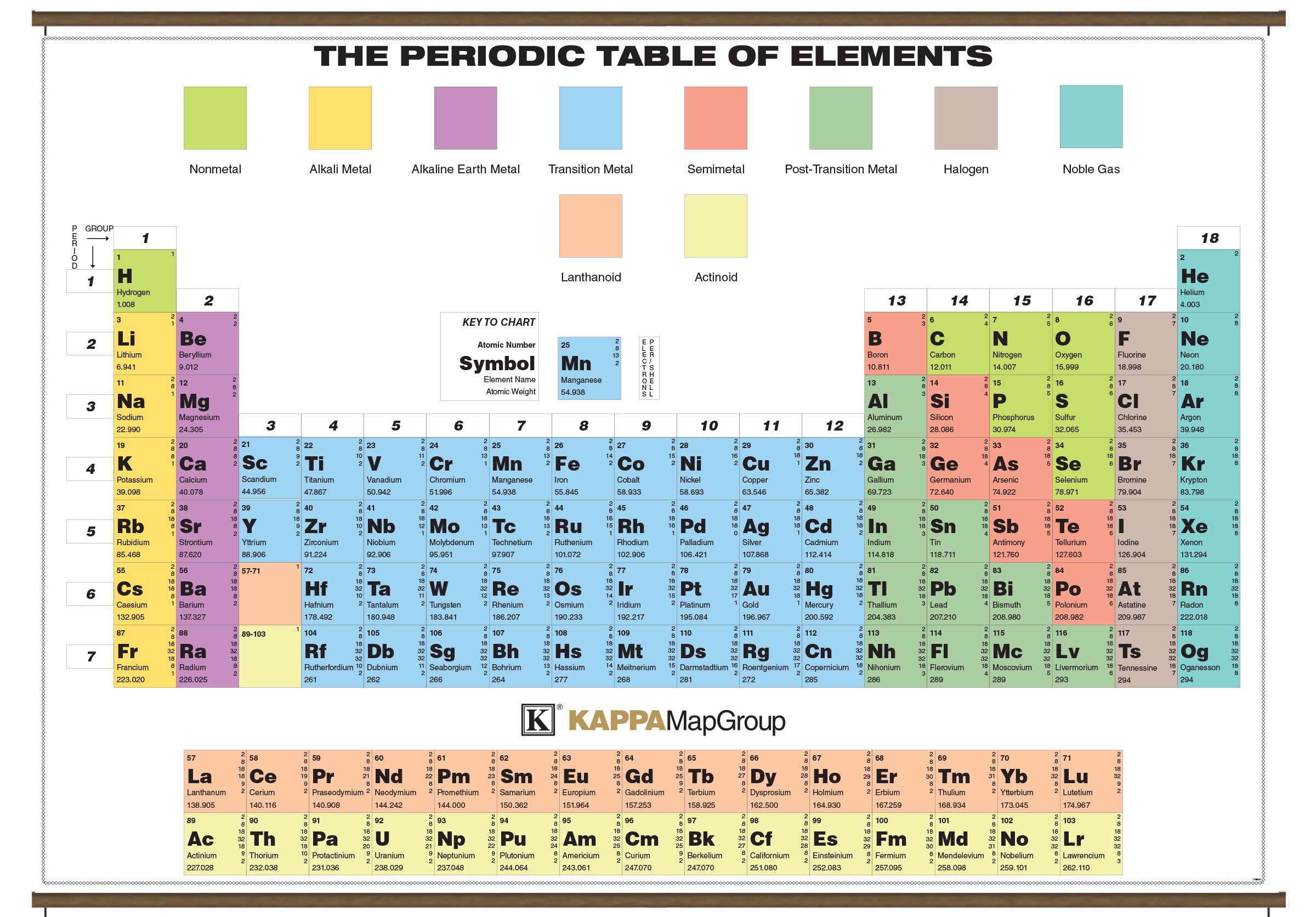
Analysis Discover the stories behind the headlines.Features Take a deeper look at the emerging trends and key issues within the global scientific community.News Stay informed about the latest developments that affect scientists in all parts of the world.Research updates Keep track of the most exciting research breakthroughs and technology innovations.Latest Explore all the latest news and information on Physics World.Geological specimens are known in which the element has an isotopic composition outside the limits for normal material.See table 1 for details of range and original paper for the atomic weight of the element from different sources.However three such elements (Th, Pa, and U) do have a characteristic terrestrial isotopic composition, and for these an atomic weight is tabulated. , indicates the mass number of the longest-lived isotope of the element. Substantial deviations in atomic weight of the element from that given in the Table can occur. Modified isotopic compositions may be found in commercially available material because it has been subject to an undisclosed or inadvertant isotopic fractionation.Value being given the tabulated value should be applicable to any normal material. Range in isotopic composition of normal terrestrial material prevents a more precise.The difference between the atomic weight of the element in such specimens and that given in the Table may exceed the stated uncertainty. See original paper for the range of these elements from different sources List of Elements with Range of Atomic Weights. See also a copy of the periodic table with atomic weights to five significant figures. See below for the elements listed in Atomic Number Order or Name order. The original paper should be consulted for full details of the variation in atomic weight and the half life of the radioisotopes quoted below.Ī number in parentheses indicates the uncertainty in the last digit of the atomic weight. For radioactive elements the isotope with the longest half-life is quoted in parenthesis. In the other lists the values quoted are those suggested for material where the origin of the sample is unknown. The standard atomic weights of twelve elements having two or more stable isotopes have variability of atomic-weight values in natural terrestrial materials. Previous values may be consulted from the 1993 table, the 1995 table, the 1997 table, the 1999 table, the 2001 table, the 2005 table, the 2007 table, the 2009 table, the 2011 table, the 2013 table, the 2015 table or the 2019 table. World Wide Web version of atomic weight data originally prepared by G. These tables are based on the 2021 table with changes from the 2019 table for the values of Ar, Hf, Ir, Pb and Yb and changes to the uncertainty for Al, Au, Co, F, Ho, Mn, Nb, Pa, Pr, Rh, Sc, Tb, Tm, and Y. 2021 Atomic Weights IUPAC Commission on Isotopic Abundances and Atomic Weights. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
